Volume 2, Issue 3, May 2015, Pages 75–81
Md. Abdullah-Al-Hasan1,![]()
![]() Md. Abdullah-Al-Mahmud2, Sultan Ahmed1, Mezbah Uddin1, S.M. Shariful Hoque Belal3, Md Humayun Kabir4
Md. Abdullah-Al-Mahmud2, Sultan Ahmed1, Mezbah Uddin1, S.M. Shariful Hoque Belal3, Md Humayun Kabir4
1Department of Microbiology and Animal Hygiene, Sylhet Agricultural University, Sylhet, Bangladesh
2Additional Veterinary Surgeon, Upazila Livestock Office, Ullapara, Sirajganj, Bangladesh
3Veterinary Surgeon, District Veterinary Hospital, Sirajganj, Bangladesh
4Bangladesh Livestock Research Institute, Savar, Dhaka, Bangladesh
| ABSTRACT | Get Full Text PDF |
A study was conducted to investigate humoral immunity level of grandparent flock following Newcastle disease virus vaccine at different intervals. The entire study was performed on 11035 chickens in Paragon grandparent farm Rajnagar, Moulvibazar during the period from July 2011 to December 2011, based on two steps. The first step included collection of sera sample from the grandparent stock broiler chickens. In the second step the humoral immunity level in chickens was measured in terms of antibody production in vaccinated chicken determined by HI test. The mean antibody titre of unvaccinated group was 4.6, 4.45, 5.02 and 4.24 in A, B, C and D line respectively. After 15 days of primary vaccination, the mean HI antibody titre was 6.9, 7.57, 7.4 and 7.9 in A, B, C and D line respectively at the age of 11 weeks and after 30 days of primary vaccination it was 6.7, 7.2, 7.2 and 6.8 in A, B, C and D line respectively at the age of 13 weeks in vaccinated group. After 15 days of second vaccination, the mean HI antibody titre was 7.1, 7.4, 7.3 and 7.5 in A, B, C and D line respectively at the age of 16 weeks and after 30 days of booster vaccination it was 4.7, 6.0, 6.1 and 6.63 respectively in A, B, C and D line at the age of 19 weeks. After 15 days of third vaccination, the mean HI titre was 7.54, 7.76, 7.0 and 7.04 in A, B, C and D line consecutively at 22 weeks of age. After 30 days of third vaccination the mean HI titre of A, B, C and D line was 6.4, 6.28, 6 and 6.05 respectively at 24 weeks of age. This mean titre was much higher after 15 days of each vaccination than that required for normal protection level. The mean HI antibody titre in chickens of unvaccinated birds were ≤ 5. After primary vaccination, the mean HI titre of vaccinated birds exhibited increased titres followed by a quick decreased titre, whereas following second and third vaccination, the HI titre increased immediately. It can be suggested that for protection of Newcastle disease second and third vaccination is essential.
Key words: Humoral immunity, broiler grandparent, Newcastle disease virus vaccine, HI test.
![]() Corresponding author.
Corresponding author.
E-mail address: dr.hasan83@yahoo.com (MAA Hasan)
How to cite this article: MAA Hasan, MAA Mahmud, S Ahmed, M Uddin, SMSH Belal and MH Kabir (2015). Determination of humoral immunity level of broiler grandparent following vaccination with Newcastle disease virus. International Journal of Natural and Social Sciences, 2(3): 75-81.
INTRODUCTION
Poultry production has a significant role in the economy of Bangladesh. It is estimated that there are approximately 138.20 millions of poultry in the country of which 75% is being reared as back yard type fed on house hold waste and crop residues (DLS, 1998). There are more than 130 hatcheries producing 3.4 millions of day-old-chicks (DOC) per-week (Rahman, 2003). Thirty thousand commercial broiler and layer farms supplies 0.26 million metric tons of poultry meat and 5210 millions of table eggs per year (GOB 1999). The investment is approximately Taka 22000 millions in the poultry sector (Rahman, 2003). But various infectious and non-infectious diseases stand on the way of advancement of poultry industries in Bangladesh. Among those, Newcastle disease (ND) appears every year in the form of epidemic, which causes 40-60% of the total mortality of poultry population in Bangladesh (Talha, 1999). ND is highly pathogenic disease of poultry and was initially reported in 1927 (Doyle, 1927). Generally, signs of ND involve depression, diarrhea, prostration, oedema of the head and wattles. The clinical signs reported in infected birds with Newcastle disease virus (NDV) vary widely but mainly depend on virulence of the virus. Birds infected with velogenic viscerotropic (vv) ND showed listlessness, increased respiration and ending with death. Diarrhea is frequently seen. Surviving birds may develop nervous signs such as muscular tremors, paralysis and torticollis (Bhaiyat et al., 1994). NDV infections are usually diagnosed by post mortem and virus isolation. Serology can be used only in non-vaccinating countries (OIE, 1996). NDV can most easily be isolated from tissue samples like lung, brain or tracheal or faecal swabs from infected birds by inoculation of eight to ten-day-old embryonated chicken eggs via the allantoic cavity (Alexander et al., 1997). Vaccination along with strict bio-security of the commercially reared backyard poultry is the way to reduce disease and thus losses resulting from infection. When designing a vaccination program, consideration should be given to the type of vaccine used, the immune and disease status of birds to be vaccinated, and the level of protection required in relation to any possibility of infection with field virus under local conditions (Allan et al. 1978). In Bangladesh, ND vaccination schedule as followed by Directorate of Livestock Services (DLS) includes administration of live lentogenic vaccine known as Baby Chick Ranikhet Disease Vaccine (BCRDV) of `F’ strain by intra-ocular inoculation during first week and 21 days old chicks followed by a live mesogenic vaccine called Ranikhet Disease Vaccine (RDV) of `M’ strain by intra-muscular route which is repeated at every six months interval (Samad et al., 2007). However, Chowdhury et al. (1981) reported that a period from day old onward to 12 days might be selected as an optimum age for primary vaccination of chicks with lentogenic B1 strain of NDV by intranasal (i/n) or oral route. But in many occasions, such a vaccination policy is not followed and the farmers resort to a vaccination schedule of their own. In breeder farm, the vaccination schedule particularly against NDV, includes a repetition of vaccination followed a closed schedule (Samad et al. 2007). This study was under taken to evaluate the immunity level of a breeder farm against NDV following vaccination and to assess the perfection of vaccination schedule.
MATERIALS AND METHODS
Study area
The study was carried out under the Paragon Grandparent Project, Rajnagar, Moulvibazar and the laboratory tests were conducted at the poultry care lab, Gazipur, Bangladesh. The study was conducted from July, 2011 to December, 2011.
Experimental chicken
A total number (11035) of experimental chickens (Hubbard classic) in Paragon grandparent farm Rajnagar, Moulvibazar was divided into two groups. Group A (comprised of thirty five birds) were kept as unvaccinated upto 7 weeks and group B comprised of eleven thousand (11,000) chickens which were vaccinated from 9 weeks of age (primary vaccination). Chicken were again divided into four experimental lines: line A (male), B (female), C (male) and D (female).
Vaccination schedule of experimental chicken
The birds were vaccinated by ND vaccine as per schedule (Table 1). Primary vaccination was done at the age of 9 weeks to all chickens of group B. The booster vaccine was administered to the birds at the age of 14 and 20 weeks. ND LaSota vaccine was used as antigen.
Table 1
Vaccination schedule against Newcastle disease.
| Age | Vaccine | Vaccine type | Dose | Route |
| 9 weeks | ND live | ND (clone30) | 1 dose | IO |
| 14 weeks | ND live | ND (Avinew) | 1dose | IO |
| 20 weeks | ND+IB live | ND+IB live(clone30+Ma5) | 1dose | IO |
| 225 weeks | ND live | ND (clone30) | 1dose | D/W |
Collection and preparation of samples
Chickens used for the collection of blood were housed separately and were not vaccinated with ND vaccine. Blood was collected aseptically either from the right jugular vein or from wing vein and collection of sera were scheduled at 7, 11, 13, 16, 19, 22 & 24 weeks of age. Following collection of blood sample, it was washed with PBS and centrifuged them thrice at 1500 rpm for 5 minutes. The supernatant was discarded and the cRBC was collected and then 2% and 0.5% cRBC was prepared in PBS for slide and micro HA tests and HI test respectively. The unused cRBC was stored at 4OC for further use.
Heamagglutination Inhibition (HI) test
Micro-plate HI test was performed using the usual standard technique (Anon, 1971) to determine the antibody level of the sera samples collected from the chicks of different lines. The test was conducted using constant 4HA unit ND virus and diluted serum (β method).
Statistical analysis
The data was analyzed statistically with the help of student’s t-test (Beri, 2005).
RESULTS AND DISCUSSION
HI antibody test
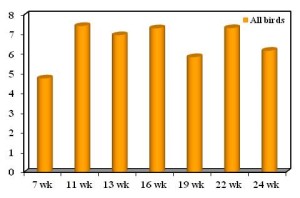
HI test was conducted to determine the antibody titres of test sera obtained from the unvaccinated and vaccinated chickens at different stages of this experiment and the titres are calculated and presented as minimum titre, maximum titre, mean titre and co-efficient of variation (CV%). The HI antibody mean titre of sera obtained from chickens belonging to vaccinated groups are presented in the Table 2 to 8. The unvaccinated group mean antibody titre was A line 4.6, B line 4.45, C line 5.02, D line 4.24 & CV was 12.7%, 16%, 9.2%, 14.2% respectively (Table 2). After 15 days of primary vaccination (ND clone30 live vaccine) the mean HI antibody titres were 6.9, 7.57, 7.4, 7.9 and CV were 10.83%, 9.36%, 11.43%, 15.36% in A, B, C and D line respectively at the age of 11 weeks (Table 3), after 30 days of primary vaccination the mean HI antibody titres were 6.7, 7.2, 7.2, 6.8 and CV were 6.75%, 11.15%, 5.34%, 11.49% in A, B, C and D line respectively at the age of 13 weeks in vaccianated group (Table 4). After 15 days of second vaccination (ND Avinew live vaccine) the mean HI antibody titres were 7.1, 7.4, 7.3, 7.5 and CV were 7.81%, 12.1%, 7.8%, 13.5% in A, B, C and D line respectively at the age of 16 weeks (Table 5) and after 30 days of booster vaccination the mean HI antibody titres were 4.7, 6.0, 6.1, 6.63 and CV were 20.34%, 14.29%, 14.19%, 17.62% respectively in A, B, C and D line at the age of 19 weeks (Table 6). After 15 days of third vaccination (ND+MA5 clone 30 live vaccine) the mean HI titres were 7.54, 7.76, 7.0, 7.04 and CV were 15.63%, 13.07%, 25.82%, 13.73% in A, B, C and D line at 22 weeks of age (Table 7). After 30 days of third vaccination the mean HI titre of A , B, C and D line was 6.4, 6.28, 6 and 6.05 respectively and CV of A, B, C and D line was 14.95%, 13.63%, 16.48% and 17.73% respectively at 24 weeks of age (Table 8). This mean titre was much higher after 15 days of each vaccination than that required for normal protection level. The mean HI titre of vaccinated birds are presented in Table 2 to 8 and demonstrated in Figure 1. The mean HI antibody titre in chickens of unvaccinated bird were ≤ 5. After primary vaccination, the mean HI titre of vaccinated birds exhibited increased titres followed by a quick decreased whereas following second and third vaccination the HI titre increased immediately.
Vaccination was performed to reduce disease and thus losses resulting from infection which is important particularly for the grandparent breeder stock. Because the immunity develops in the grandparent stock is transferred to baby chick through egg yolk and such maternal antibody is helpful to protect the chicken from the infectious disease in their early life. It was found that the higher the antibody level in grandparent breeder stock the better maternal immunity is conferred in baby chick. Heller et al. (1977) conducted an experiment on the transfer of Newcastle serum antibody from the laying hen to the egg and chick and reported that hens with antibodies to Newcastle disease virus passed these on to their progeny via the egg yolk.
Table 2
HI titres of sera collected from unvaccinated chickens.
| Line | Age | Samples | Max Titre | Mini Titer | Mean titre | CV% |
| A Line | 7w | 5 | 7 | 4 | 4.6 | 12.7 |
| B Line | 10 | 7 | 3 | 4.45 | 16 | |
| C Line | 10 | 6 | 3 | 5.02 | 9.2 | |
| D Line | 10 | 7 | 3 | 4.24 | 14.2 |
Table 3
HI titres of sera collected from chicken 15 days post- primary vaccination.
| Line | Age | Routeof vaccination | No. of sample | Max. Titre | MiniTitre | MeanTitre | CV% |
| A line | 11wk | IO | 5 | 8 | 5 | 6.9 | 10.83 |
| B line | 14 | 8 | 6 | 7.57 | 9.36 | ||
| C line | 10 | 8 | 5 | 7.4 | 11.43 | ||
| D line | 47 | 9 | 5 | 7.9 | 15.36 |
Table 4
HI titres of sera collected from chicken 30 days post- primary vaccination.
| Line | Age | Route of vaccination | No. of sample | Max. Titre | MiniTitre | MeanTitre | CV% |
| A line | 13 wk | IO | 5 | 8 | 6 | 6.7 | 6.75 |
| B line | 14 | 9 | 6 | 7.2 | 11.15 | ||
| C line | 10 | 9 | 6 | 7.2 | 5.34 | ||
| D line | 47 | 10 | 5 | 6.8 | 11.49 |
Table 5
HI titres of sera collected from chicken 15 days after booster vaccination.
| Line | Age | Route of vaccination | No. of sample | Max. Titre | MiniTitre | MeanTitre | CV% |
| A line | 16 wk | IO | 5 | 8 | 6 | 7.1 | 7.81 |
| B line | 14 | 8 | 6 | 7.4 | 12.1 | ||
| C line | 10 | 9 | 7 | 7.3 | 7.8 | ||
| D line | 47 | 8 | 6 | 7.5 | 13.5 |
Table 6
HI titres of sera collected from chicken 30 days after booster vaccination.
| Line | Age | Route of vaccination | No. of sample | Max. Titre | MiniTitre | MeanTitre | CV% |
| A line | 19wk | IO | 5 | 6 | 4 | 4.7 | 20.34 |
| B line | 14 | 8 | 4 | 6.0 | 14.29 | ||
| C line | 10 | 7 | 5 | 6.1 | 14.19 | ||
| D line | 47 | 7 | 3 | 6.63 | 17.62 |
Table 7
HI titres of sera collected from chicken 15 days after second booster vaccination.
| Age | Route of vaccination | No. of sample | Max. Titre | MiniTitre | MeanTitre | CV% | |
| A line | 22wk | IO | 5 | 9 | 5 | 7.54 | 15.63 |
| B line | 14 | 9 | 6 | 7.76 | 13.07 | ||
| C line | 10 | 9 | 4 | 7.0 | 25.82 | ||
| D line | 47 | 10 | 5 | 7.04 | 13.73 |
Table 8
HI titres of sera collected from chicken 30 days after second booster vaccination.
| Age | Routeof vaccination | No. of sample | Max. Titre | MiniTitre | MeanTitre | CV% | |
| A line | 24 wk | IO | 5 | 7 | 5 | 6.4 | 14.95 |
| B line | 14 | 8 | 5 | 6.28 | 13.63 | ||
| C line | 10 | 8 | 4 | 6.0 | 16.48 | ||
| D line | 47 | 8 | 4 | 6.05 | 17.73 |
Figure 1
Histogram showing the level of average mean HI titres of of chicken at different time points.
Aryal et al. (2003) conducted a study to detect the persistence of maternally derived ND specific antibody in progeny from vaccinated parent stock and to examine the level of NDV specific antibody produced in mature chicken. The authors found that the levels of antibody were significantly different in the different age groups of the same breed called Hisex 9. Islam et al. (2003) reported that there was a wide variation in the maternally derived antibody in day-old chicks where the –upper and lower titre was 753 and 60 respectively and the mean titre was 247.9.
In Bangladesh the schedule of vaccination against Newcastle disease as followed by DLS includes the administration of live lentogenic vaccine (BCRDV) of F-strain by oral or i/n inoculation during the 1st week and 21days of age. Day-old chick is vaccinated by a live mesogenic vaccine (RDV) of M-strain by i/m which is repeated at every six months interval. However, Kafi (2003) reported that vaccination schedule consisting of a live lentogenic vaccine- followed by an inactivated or killed vaccine conferred higher and durable level of immunity. For this reason, poultry breeders usually used a live lentogenic vaccine for the chick followed by an inactivated vaccine to obtain the better immune response. Amin et al. (1987) reported that serological response of chickens to NDV either from natural infection or vaccination was manifested by the appearance of both HI and VN antibody. It was reported that both HI and VN antibodies though follow a similar course but VN antibody persists longer in relatively high titre (Brandly et al., 1947; Hanson, 1972 and Haplin, 1978). It is further mentioned that HI test provides indirectly a measurement of the ability of serum from an exposed bird to neutralization of NDV, inhibited agglutination of chicken RBC by NDV, whereas SN test indicated directly the ability of serum to neutralize the infective property of NDV and therefore provided more precise information about the immune response (Hanson, 1964). Amin et al. (1987) observed that sera samples containing HI titre of 80 or more indicated status of birds equivalent to neutralizing effect of homologues virus where as sera sample containing 40 or less did not posses such properties. Allan and Gough (1974) showed a HI titre of 23 indicated the birds would be protective against fatal ND if challenged. In the present study, when A, B, C and D line birds were vaccinated at 9 weeks of age and sera were collected at 11 weeks of age, the mean antibody titre showed 6.9, 7.57, 7.4, 7.9 respectively (Table 3) and every line is different from each other. The reasons might be due to the variation of feeding, management or individual variation. Haplin (1971) mentioned that individual bird might have difference in the elucidation of HI response. The similar phenomenon of non-responsiveness to vaccination by a small number of birds and irregularity in HI antibody response was also observed by a number of investigators (Lancaster, 1966; Ahmed et al., 1967 and Hervert, 1970). After primary vaccination, the mean HI titre (Table 3) of vaccinated birds showed increased titre followed by decreased titre (Table 4) whereas following second vaccination, the mean titre (Table 5) increased followed by decreased titre (Table 6) and in third vaccination, the HI titre increased (Table 7) then decreased (Table 8). Chowdhury et al. (1982) reported that HI antibody titre begins to decline after 3 weeks of vaccination. Ali et al. (1978a) reported that intranasal inoculation of K-strain ND vaccine at 4 week-old birds gave 100% immunity as indicated by HI titre and challenged test. However, the level of immunity was recorded to be less than 80% after 23 weeks of vaccination. When ND live vaccine (ND clone30) administered to grandparent birds at 9 weeks of age the HI antibody mean titre in A, B, C, D line were 6.9, 7.57, 7.4, 7.9 respectively (Table 3) at the age of 11 weeks. This mean titre is higher than that required for normal protection level (titre 6.5). Bird vaccinated twice with ND clone-30 vaccine and at 120 days with oil adjuvanated killed vaccine showed higher level of antibody production (Rahman et al., 2001). Considering the result of the study it is indicated that live vaccine produced higher level of antibody titre. After primary vaccination, the mean HI titre of vaccinated birds showed increased titres and started to decline whereas following second and third vaccination the HI titre increased immediately. It can be concluded that for better protection from Newcastle disease second and third vaccination (Booster vaccination) is essential. It may be also concluded that vaccination with a ND live vaccine provoked a dependable level of immunity and such a high level of antibody as were observed in this study could be useful for breeder birds to provide better protection of their chick by transferring high level of maternal antibody.
REFERENCES
Ahmed AAS, Roda IM, Abbasi KH and EI Sisi MA (1967). Studies on the immune response to vaccination against Newcastle disease in UAR, Journal of Veterinary Science, UAR 4: 115-133.
Alexander DJ, Manvell RJ, Lowings JP, Frost KM, Collins MS, Russell PH and Smith JE (1997). Antigenic diversity and similarities detected in avian paramyxovirus type I (Newcastle disease virus) isolates using monoclonal antibodies. Avian Pathology, Oxfordshire: Cartax Publishing Company. 26: 399-418.
Ali B EIh (1978). Response to chicks to vaccination with K-strain of Newcastle disease virus under field conditions. Indian Veterinary Journal, 55: 508-512.
Ali WH, Lancaster JE and Toth Q (1978). Newcastle disease vaccines, their production and use. Chapter 3. Selection of the vaccine seed strain, pp. 1018. Food and Agricultural Organization of the United Nations, Rome.
Allan WH and Gough RE (1974). A standard haemagglutination inhibition test for ND (2) vaccination and challenged. The Veterinary Record, 95: 147-149.
Allan WH, Lancaster JC and Toth B (1978a). Newcastle disease vaccines, their production and use. Food and Agricultural Organization (FAO), Animal Production Series, No. 10. FAO. Rome.
Amin MM, Sarker AJ, Ali MR and Saifuddin M (1987). Investigation of some parameters for measuring immune responses following vaccination with ND. Bangladesh Veterinary Journal, 21: 33- 42.
Aryal P, Hossain KM, Ahasan MM, Islam KMD, Billah MM, Islam ME, Mehedi M and Mitra S (2003). Seroprevalence of Newcastle disease virus specific antibody in chicken. Journal of Animal and Veterinary Advances, 2: 366- 371.
Bhaiyat MI, Ochiai K, ltakura C, Islam MA and Kida H (1994). Brain lesions in young broiler chickens naturally infected with a mesogenic strain of NDV. Avian Pathology, 23: 693-708.
Brandly CA, Hanson RP, Lewish SH, Winslow NS, Hoit HH, Pritchard WR and Nerlinger CM (1947). Variables and correlation in laboratory procedures for ND diagnosis. Cornell Veterinary, 37: 324-336.
Chowdhury SI, Chowdhury TIMFR, Sarker AJ and Amin MM (1981). Determination of an optimum age for primary Newcastle disease vaccination of chicks having maternal antibody. Bangladesh Veterinary Journal, 15: 19-17.
Chowdhury SI, Chowdhury TIMFR, Sarker AJ, Amin MM and Hossain WIMA (1982). Studies on Newcastle disease in Bangladesh. A research report. 2. The role of residual maternal antibody on immune response and selection of an optimum age for primary vaccination of chicks. pp. 12- 22.
Campbell RSF (1986). The pathogenesis and pathology of avian respiratory infection. Veterinary Bulletin, 56: 521-543.
Deleeuw O and Peeters B (1999). Complete nucleotide squence of Newcastle disease virus: evidence for the existence of a new genus within the subfamily Paramyxovirinae. Journal of General Virology, 80: 131-136.
DLS (1998). Livestock population. Directorate of Livestock Services, Khamarbari Road, Farmagate, Dhaka 1215.
Doyle TM (1927). A hit hert unrecorded disease of fowls due to a filter passing virus H. Comparative Pathology and Therapy, 40:144-169.
GOB, 1999. The Reported on the Economic Survey of Bangladesh, Ministry of Finance, Government of Peoples’ Republic of Bangladesh, Dhaka.
Hanson RP (1964). Newcastle disease: An evolving pathogen. Univ. Wisconsin press, Madison. (Cited from Amin MM, Sarker AJ, Ali MR and Saifuddin M (1987). Investigation of Some parameters for measuring immune respones following vaccination with Newcastle disease. Bangladesh Veterinary Journal, 21: 33-42.
Haplin B (1971). Patterns of Animal Disease. Chapter-8, Immunity and Epidemiology. The English Language Book Society and Bailliere Tindal. London. pp. 151-157.
Haplin B (1978). Patterns of Animal Disease. Chapter-8, Immunity and Epidemiology. The English Language Book Society and Bailliere Tindal. London. pp. 151-157.
Hervert WJ (1970). Veterinary Immunology, Edinburg; Livingstone. (Cited from Saifuddin M, Sarker AJ, Amin MM and Rahman MA (1986). Studies on the efficacy of ND vaccines and their vaccination schedule. Bangladesh Veterinary Journal, 20: 67-75.
Heller ED, Nathan DB and Perck M (1977). The transfer of Newcastle serum antibody from the laying hen to the egg and chick. Research in Veterinary Science, 22: 376-379.
Islam MR, Huque QME, Giasuddin M, Alam J and Rahman MM (2003). Assessment of maternally derived antibody of commercial flock against Newcastle disease. Proceeding of 3rd International Poultry show and seminar, Bangladesh China Friendship Conference Centre, Dhaka, Bangladesh, 28th February-2nd March, 2003.
Kafi MA (2003). Vaccination studies on use of Ranikhet inactivated vaccine combined with V4HR or BCRDV. M.S. Thesis, Department of Microbiology and Hygiene, BAU, Mymensingh.
Lancaster JE (1966). Newcastle disease. A Review, 1926-64. Canada Department of Agric., Ottawa, Canada.
Lancaster JE (1981). Newcastle disease. In: Gibba E.P.J. (Ed.) Virus Disease of Food Animals. Vol. II, Disease Monographs, Academic Press, New York, pp. 433-465.
Office International Des Epizooties (OIE) (1996). Newcastle disease. In: Manual of standards for diagnostic tests and vaccine. 3rd Ed. 0IE, Paris, pp. 161-169.
Rahman MM (2001). Evaluation of humoral immunity against newcastle disease virus in vaccinated chick. M.S. Thesis, Department of Pathology, BAU, Mymensingh.
Rahman MM (2003). Growth of poultry industry in Bangladesh, Poverty alleviation and employment opportunity. Proceeding of the 3rd international poultry show and seminar, Bangladesh China Friendship Conference Center, Dhaka, Bangladesh, 28th February-2nd March, 2003.
Samad MA, Kafi MA, Amin MM and Gani MO (2007). Investigation on the immunity level of breeder flocks following Newcastle disease virus vaccine. Bangladesh. Journal Veterinary Medicine, 5: 15–18.
Talha AFSM (1999). Pathology of poultry diseases occurring in Mymensingh. M.S. Thesis, Department of Pathology, Faculty of Veterinary Science, BAU, Mymensingh.